NHS England has set ambitious targets to improve stroke survival rates. Its strategy includes wider access to mechanical thrombectomy, a procedure used to remove a blood clot in a patient’s brain. Utilising a small device known as a stent retriever, a tiny stent-shaped medical device that is inserted into the blocked artery in the brain, the device ensnares the blood clot and removes it from the body.
In April 2018, the NHS will begin commissioning 24 neuroscience centres in the UK to receive reimbursement and funding to perform mechanical thrombectomy procedures. Currently only a few hundred patients a year receive the treatment and just a handful of hospitals in the UK offer it, despite its proven benefits.
The procedure can produce remarkable results, with patients who may have otherwise been disabled, capable of leading independent lives. Annually, about 8,000 people who have an ischemic stroke may benefit from the expansion in the number of hospitals offering mechanical thrombectomy.
This new approach aims to capitalise on the rapid technological innovation in the field of stroke care, bringing new hope to thousands of men and women who suffer strokes every year, improving the quality of life for stroke survivors.
At the forefront of this change in stroke care is Stryker, one of the world’s leading medical technology companies. Emerging evidence from the Stryker sponsored DAWN TrialTM on mechanical thrombectomy and its benefits in terms of recovery from stroke and quality of life are providing important clinical data and supporting other initiatives focused on the development of stroke-care pathways around the world, including the UK.
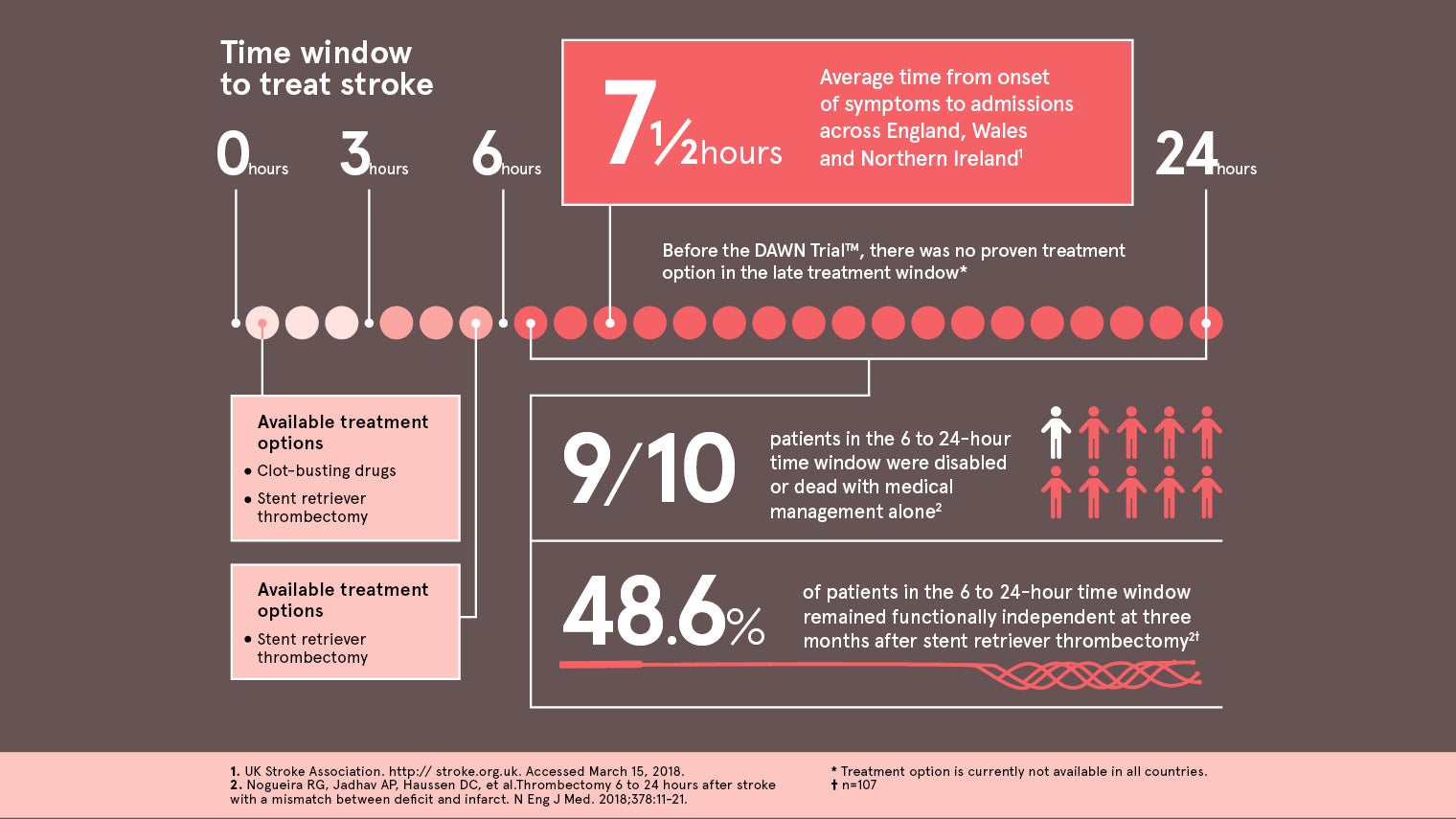
About 48 per cent of patients treated with the Trevo device in the six to twenty four-hour window were functionally independent at three months, compared with only 13 per cent of patients treated with medical management alone. Carlos Pena, director of neurological and physical medicine at the FDA, said: “Healthcare providers and their patients now have better tools for treating stroke and potentially preventing long-term disability.”
The DAWN TrialTM compared 107 patients treated with the Trevo Retriever and medical management to 99 patients who received only medical management. In the DAWN TrialTM nine out of ten patients who did not receive thrombectomy were either dead or disabled 90 days after their stroke.
Until now, mechanical thrombectomy devices like the Trevo Retriever were only cleared for use up to six hours from symptom onset, leaving a significant population of stroke patients ineligible to receive treatment because they arrived at a hospital that doesn’t treat stroke or outside the treatment window. The DAWN study and its findings open up that potential treatment window for patients up to 24 hours. We are living our mission to make healthcare better. However, every minute still matters in stroke treatment. It is still imperative for all caregivers in the stroke treatment pathway, from emergency medical services to emergency room physicians to neurologists and interventionalists who do the procedure, to continually improve the speed in which they can deliver fast and efficient treatment of stroke. With stroke, every minute counts.
For more information please visit stryker.com

