A new medical technology, which has the potential to revolutionise the chest-pain pathways in hospitals around the world, is about to go live, backed by a major clinical study and successful private funding rounds.
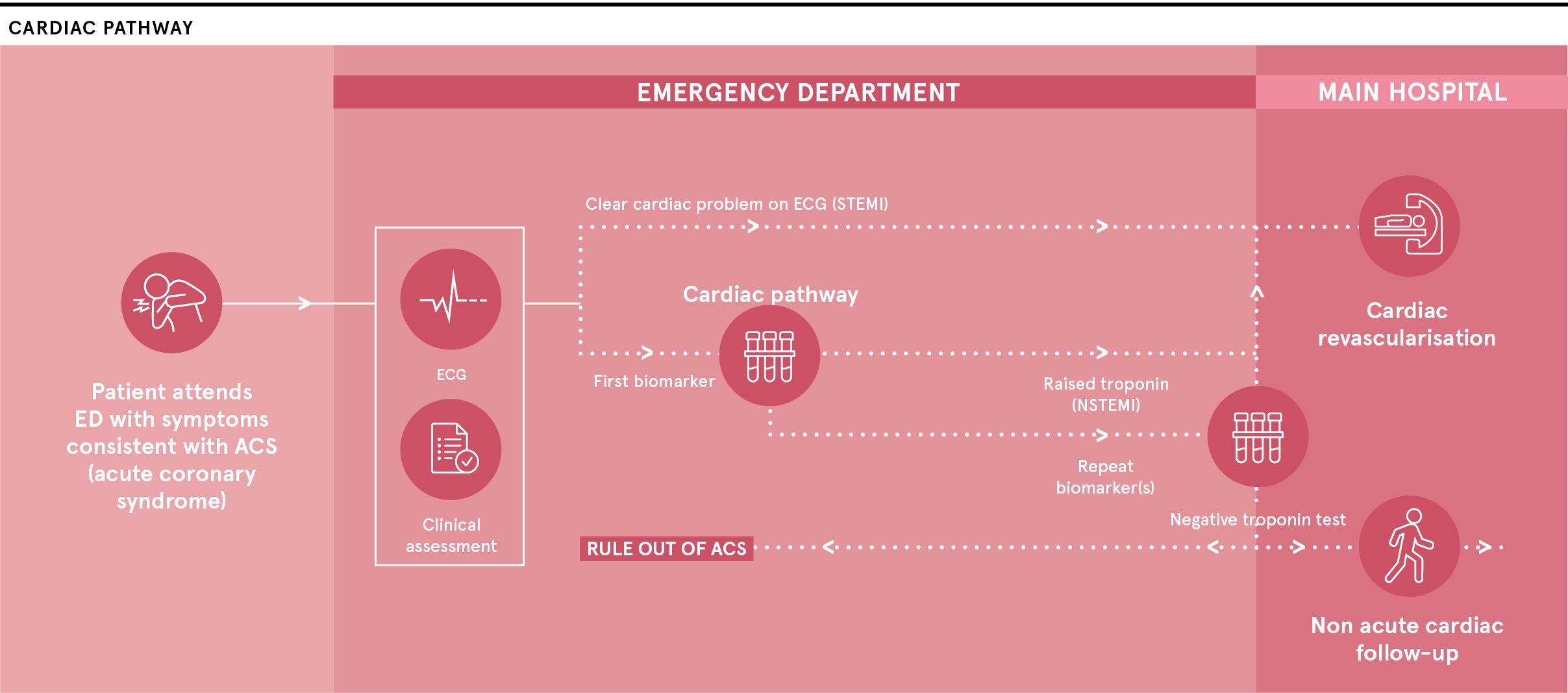
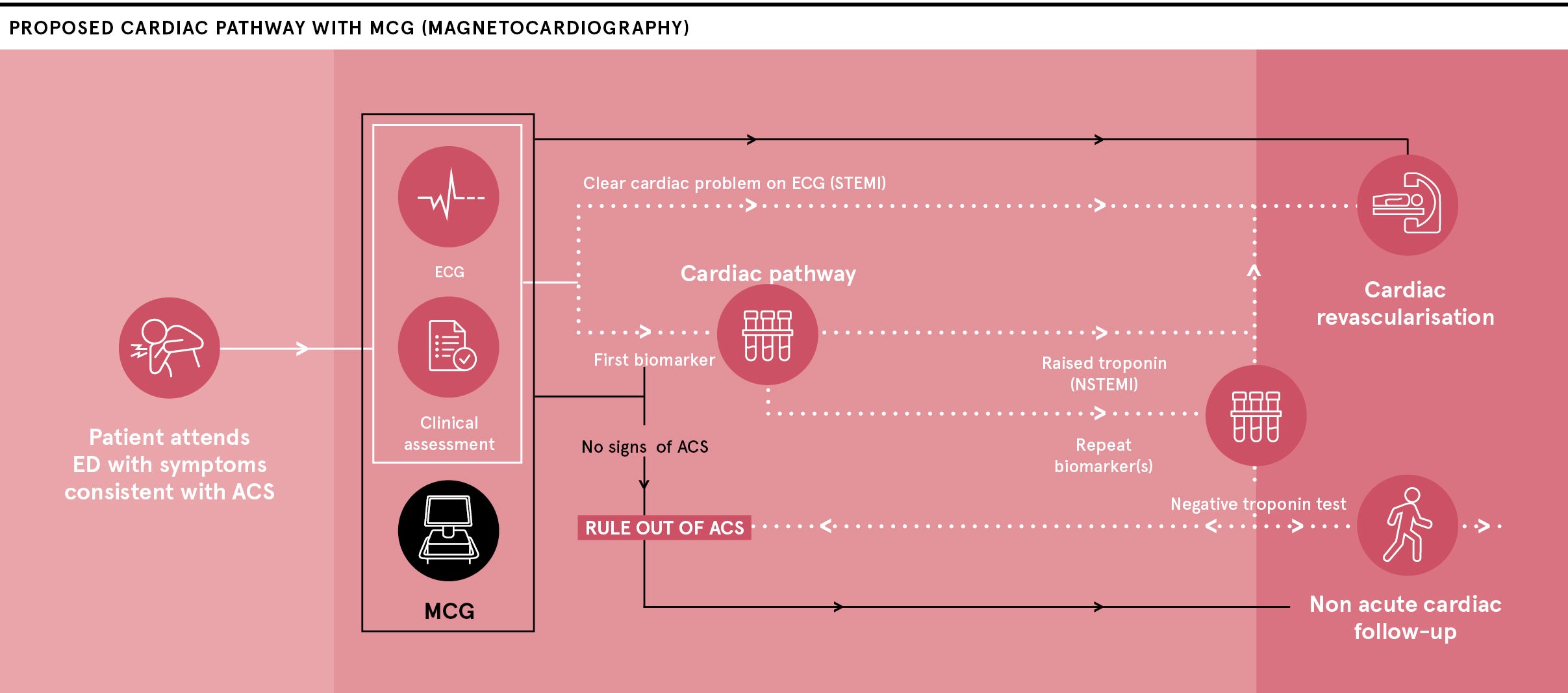
Creavo Medical Technologies is at the pivotal moment in a five-year journey to develop its award-winning Corsens® system, which is intended to act as an aid for physicians to give an early all-clear to a number of people who attend hospitals with chest pain and are currently forced to complete a lengthy and anxious testing pathway.
The technology, which applies principles of physics to measure the heart’s magnetic signals, means that some of these chest-pain patients can be reassured they are not having a heart attack and can return home rather than spending up to eight hours in a hospital.
“The target audience are people who have been completely well one moment then they suffer chest pain which they assume is a heart attack. They are rushed into hospital and are naturally fearful,” says Creavo chief executive Steve Parker, who had more than 40 years’ experience in giant US medical technology companies before launching the startup, a spin-out from Leeds University, in 2014.
“If we can identify those not at risk at an early stage, we put both their minds at rest and free up resources in the emergency department for the people who really do need them. This is a patient, economic and technology story that comes together to make a very effective business model.”
The technology, which Creavo has packed into a portable unit, can assess the heart’s condition within minutes by a non-invasive scan, delivered encouraging results in early studies and is about to receive the results of a large study at five busy UK hospital emergency departments.
There is a large amount of interest in the technology with many saying it has the potential to be a game-changer.
The company is just embarking on its latest funding round with continued support from its existing private healthcare investors, including the IP Group. It has just moved into a new base in Coventry and is gearing up for production of the compact device, which can be wheeled to a patient’s bedside and operated by healthcare professionals.
Creavo has an impressive medical advisory board. “We have great support from our medical advisory board. They are positively engaged and that is a great indication of the excitement it is generating,” says Mr Parker. “We have not been short of investment and support because we have the right people and have a very engaging story; most people will know someone who has been through that hospital process. We have been
supported very strongly by our investors, which has enabled us to grow the UK study very quickly. They are committed to our future.”
The UK study results are expected towards the end of the year and will coincide with a planned European market launch, followed by further studies and a US launch towards the end of 2020.
This is a patient, economic and technology story that comes together to make a very effective business model
“The pure business model is that patients who go through the pathway, but don’t have a problem, are expensive to hospitals,” Mr Parker adds. “Even using conservative numbers, we expect to have a huge impact. A major cost is incurred assessing people at low risk of heart attack by putting them through the current chest-pain pathway.
“The really exciting element is that this is a platform technology with multiple applications and £20 billion of business opportunities, but we are resolutely focused on getting Corsens to market. We have planned to achieve regulatory clearance on our device in the second half of this year to coincide with publication of clinical trial data, which will really start our commercial journey in Europe. We will also be closing recruitment in a US study around the third quarter of 2019, which starts us on the road to a US launch by the end of 2020.”
IP Group has been involved with the device since its inception and is the company’s longest-running and biggest shareholder. Dr Simon Graindorge, a partner in its life sciences and healthcare division, says: “I have been investing in early-stage technologies for 15 years and have never been involved in a technology that has such a widespread need as this, and is universally understood and endorsed by clinicians who understand the problem.
“Every clinician you talk to cuts you off at about the second sentence because they understand exactly what the problem is; they deal with it daily and understand how this has the potential to change what they do. The clinical support has been powerful and they have a strong management team that is doing a great job.”
IP Group came on board after Dr Graindorge met Professor Ben Varcoe of the University of Leeds School of Physics and Astronomy, who suffered heart trouble and was told by his cardiologist at the end of a busy clinic: “At last, someone with a genuine problem.”
The burden of pushing a procession of patients through cardiac testing and then follow-up clinic appointments was explained, and Professor Varcoe began applying his knowledge of quantum electrodynamics to map the electrical signals of a healthy heart.
Professor Varcoe, who is Creavo’s chief scientific officer, discovered abnormal patterns in the magnetic fields reflect a loss of oxygen to the heart muscle (ischaemia), indicating the need for further tests and treatment, while a heart free from ischaemic issues emits a different set of signals that are picked up by the device.
“We specialise in investing in early-stage projects deriving from university research,” adds Dr Graindorge. “This is a genuine platform that can be used in different areas, aside from its first application in the emergency setting. If the company can achieve its goals over the next two to three years, it will be in a fantastic place.”
Creavo has just celebrated its fifth anniversary with a move to new premises future-proofed for its expansion and the commercialisation of the Corsens device for which it has already secured European and Middle-East distribution partners.
The company and its staff have won industry accolades at the Institute of Engineering and Technology Innovation Awards, which celebrate the very best in new innovations in science, engineering and technology, showcasing entries from around the world. It followed up with the Institute of Physics’ Start Up Award in 2018.
“The awards are great recognition for the work we have achieved as a young company with a great business idea with huge potential that is founded on a physics invention,” says Mr Parker. “We are driven by a desire to get these products to the frontline of healthcare where they can make a significant difference to patients, doctors and national health systems.
“This is a British invention, funded predominantly by British investors and we have created 50 high-calibre jobs. It talks to the levels of innovation in the UK, the investment, the tax credits and all the elements that have helped us, and demonstrates the UK market is very strong in supporting the medtech business. It is a great British success story.”
For more information please visit creavomedtech.com